Cloud-Based Clinical Research Software
This case study explores how Closeloop designed and delivered Vestigo — a fully custom, cloud-based platform for the McCreadie Group — empowering Investigational Drug Services to manage protocol budgets, checklists, drug tracking, and clinical trial workflows in one seamlessly integrated solution.
The McCreadie Group provides a cutting-edge software solution called Vestigo for Investigational Drug Services (IDS). Vestigo stands as a web-based platform utilized by Investigational Drug Services to effectively oversee investigational drug products.
This comprehensive platform facilitates the tracking of both significant and intricate details, encompassing trial statuses, study teams, patient enrollments, IRB expirations, and more.
Vestigo seamlessly interfaces with OnCore, Research Pharmacists, EMR Systems, and Clinical Trial Management Systems (CTMS), making it the go-to platform for clinical trial management.

Operational Gaps in Protocol Management
Upon thorough analysis of the current process, business requirements, and workflow within Vestigo, it became evident that certain features, flows, and business aspects needed enhancement to ensure heightened security and transparency within the system.
Manual Budget Creation
The Investigational Pharmacy relied on Excel to compile fees based on study phase, drug preparation, storage, and administration — demanding significant time to create, manage, and keep files updated.
No Active Study Linkage
There was no mechanism to link the study once it became active, impeding monitoring of provided costs against the actual expenses incurred during the trial lifecycle.
Fragmented Protocol Checklists
Sites resorted to disparate Excel or Word solutions to track and allocate tasks pertaining to protocol development — an unwieldy method that failed to integrate with Vestigo's broader solution.
Inconsistent Workflows
Tasks such as budgeting, Site Qualification, drug builds in EMR systems, and Vestigo build verification lacked a unified and standardized tracking mechanism across sites.
Security & Transparency Gaps
The existing process lacked the security controls and visibility required to manage investigational drug products at the level expected by regulatory and institutional standards.
Integrated Clinical Workflow Automation
Closeloop addressed the core operational challenges by delivering two tightly integrated solutions within the Vestigo platform — Protocol Budget and Protocol Checklist — each designed to standardize, automate, and bring transparency to the clinical trial workflow.
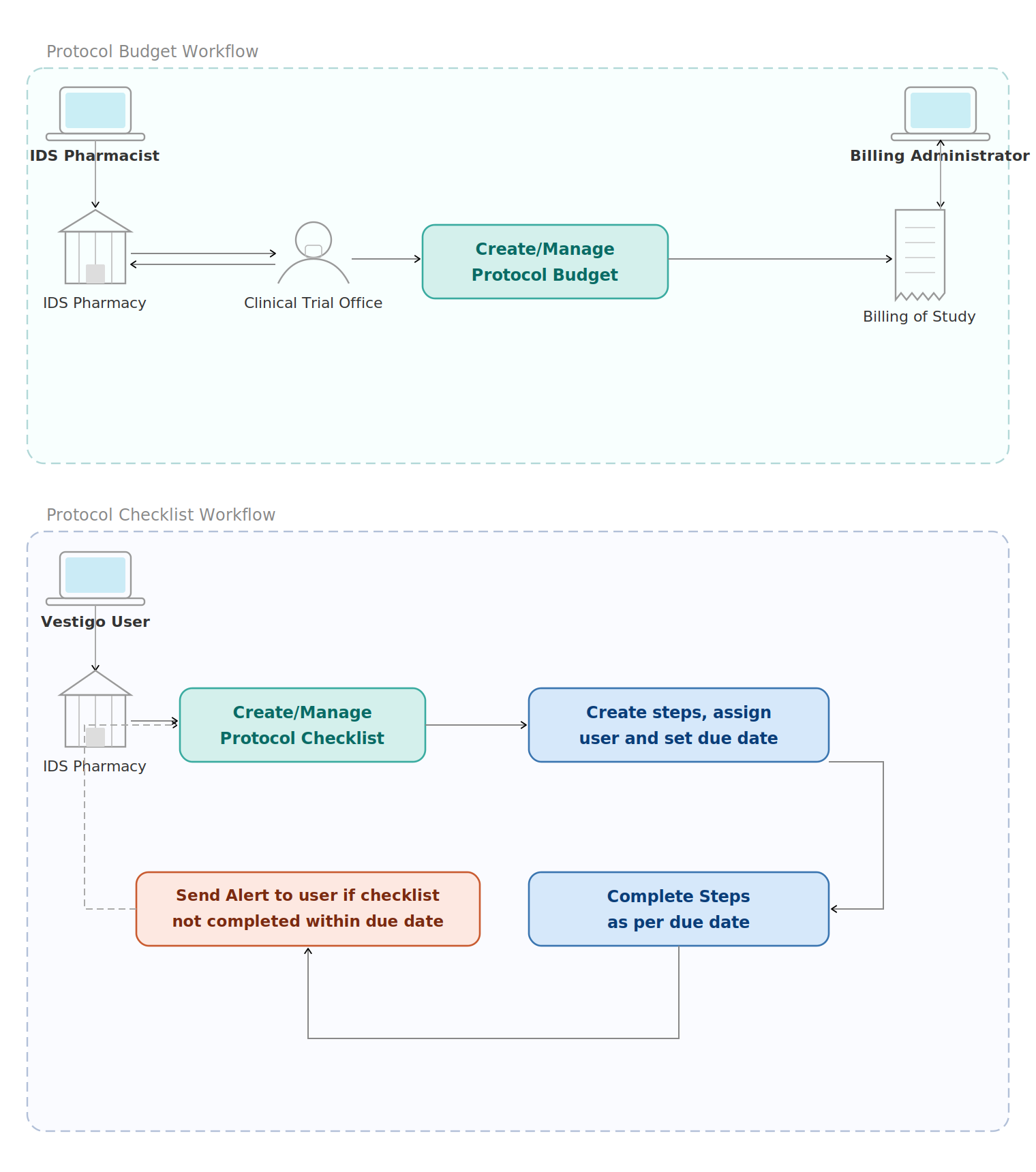
Protocol Budget
Provided the Investigational Pharmacy with an electronic solution within Vestigo to build consistent and repeatable budget templates. Pharmacy teams can now use these templates when creating protocols, eliminating the need to reconfigure fees each time a new budget is approved.
- Reusable budget templates for studies
- Reliable repository of all budgets
- Visibility into quantity and fee structures
- Linked to active protocols for cost monitoring
Protocol Checklist
Implemented a Protocol Checklist that leverages templates to provide a consistent method of managing and reporting on the status of protocol tasks — from Site Qualification and budgeting (pre-approval) to drug builds in EMR and Vestigo build verification (while live).
- Template-driven, standardized checklists
- Common practice across sites and facilities
- Assign users and set due dates per task
- Automated alerts for overdue checklist items
Development & Solution Architecture
Closeloop designed, developed, deployed, and maintained the McCreadie Group's Vestigo software — a fully custom software platform for supporting all aspects of Vestigo's business. Vestigo is integrated with 3rd party vendors and includes a full set of APIs for external partners to connect.
Designed and built an intuitive user interface tailored to pharmacy and clinical trial workflows.
Architected a scalable, modular system capable of supporting multiple integrations and future expansions.
Seamless integration with OnCore, EMR Systems, and Clinical Trial Management Systems (CTMS).
Rigorous QA testing, user acceptance testing, and staff training prior to final deployment.
Platform Capabilities
- Full-cycle protocol management from initiation to closure
- IRB expiration tracking and renewal alerts
- Patient enrollment and study team management
- Investigational drug product inventory oversight
- Budget templating and cost reconciliation
- API suite for external partner connectivity
Technical Architecture
Tech Stack
 ASP.Net
ASP.Net
 VB .Net
VB .Net
 Database SQL
Database SQL
 JavaScript
JavaScript
 BackBone JS
BackBone JS
Business Impact and Results
Closeloop developed and delivered two-factor authentication and an automated removal process, transforming the way the McCreadie Group's platform handles security and program administration.
Approximately a 45% reduction in daily support tickets related to password resets.
Improved capability for Residency Program Directors (RPD) to manage programs effectively.
Streamlined password recovery process for residents, enhancing overall user satisfaction and security measures.
Client Value & Feedback
Client feedback reflects not just satisfaction with technical execution, but appreciation for the strategic partnership and business outcomes achieved.
"Closeloop designed, developed, deployed, and maintained our Vestigo software — a fully custom platform for supporting all aspects of our business. The platform was conceptualized and delivered within 4–5 months, including frontend UI, solution architecture, coding, QA, training, and final deployment. Their team truly understood our clinical research workflows and translated them into a system that works."
— McCreadie Group, Vestigo Product TeamFrequently Asked Questions
No questions match your search.
Investigational pharmacies that rely on Excel to compile protocol budgets must manually configure fees every time a new study is initiated — factoring in study phase, drug preparation, storage, and administration costs from scratch. This process is time-consuming, error-prone, and impossible to standardize across sites or studies. There is also no live linkage between the budget and the active protocol, meaning teams have no visibility into how approved costs compare against actual expenses as the trial progresses. Closeloop Technologies solved this by building a reusable, template-driven budget module directly inside the clinical platform — eliminating manual recreation and enabling real-time cost monitoring against active protocols.
Closeloop handled the complete end-to-end delivery — including requirements analysis, solution architecture, frontend UI design, backend development, third-party integrations, QA testing, user training, deployment, and ongoing maintenance. The platform was built on ASP.Net, VB.Net, SQL, JavaScript, and Backbone.js, with a full API suite enabling connectivity to external partners. The entire platform — covering frontend UI, solution architecture, coding, QA, training, and final deployment — was conceptualized and delivered within 4–5 months, demonstrating the team's ability to move fast without compromising on quality or clinical workflow accuracy.
A Protocol Checklist is a structured, template-driven task management system that tracks the status of every protocol development activity — from pre-approval tasks like Site Qualification and budgeting, to live-phase activities like drug builds in EMR systems and Vestigo build verification. Without it, sites resort to disconnected Excel or Word documents that are impossible to standardize across facilities. A properly implemented checklist assigns tasks to specific users, sets due dates, sends automated alerts for overdue items, and gives program managers a real-time view of protocol readiness. Closeloop Technologies built this capability natively into the Vestigo platform, replacing fragmented manual tracking with a single standardized system across all sites.
Investigational Drug Services operate at the intersection of multiple systems — EMR platforms for patient data, CTMS for trial management, and OnCore for research coordination. When these systems operate in silos, pharmacy teams must re-enter data across platforms, increasing the risk of errors, delays, and compliance gaps. Tight integration ensures that drug builds, patient enrollment status, IRB expiration dates, and trial updates flow automatically between systems — giving all stakeholders a consistent, real-time view without manual data reconciliation. Closeloop Technologies architected Vestigo's integration layer to connect seamlessly with OnCore, EMR systems, and CTMS, enabling a unified clinical workflow across all connected platforms.
Excel-based budget management in clinical research introduces significant operational and compliance risks — files go out of sync, version control is unreliable, there is no audit trail for changes, and there is no linkage between the budget document and the live protocol it governs. When a study becomes active, teams have no automated mechanism to monitor actual costs against approved fees, leaving financial oversight entirely dependent on manual reconciliation. For regulated environments like investigational pharmacies, this level of opacity is both operationally inefficient and potentially non-compliant. Closeloop Technologies replaced this fragile approach with an electronic budget module that is fully integrated into the clinical platform and linked directly to active protocols.
Protocol development involves dozens of sequential and parallel tasks — each with deadlines, responsible parties, and downstream dependencies. Without automated alerts, overdue tasks go unnoticed until they create a bottleneck or compliance gap. Automated alerts notify assigned users when tasks are approaching or past their due date, keeping the entire team accountable without requiring manual follow-up from program managers. Closeloop Technologies built this alerting and assignment logic into the Vestigo Protocol Checklist feature, ensuring that no task falls through the cracks regardless of how many protocols or sites are being managed simultaneously.
A fully custom clinical platform covering core workflow modules, third-party integrations, QA, training, and deployment typically takes 4–6 months for a focused initial scope, and 12–15 months for a comprehensive multi-module system with a full API suite and integrations to multiple external clinical systems. Closeloop Technologies delivered the initial Vestigo platform within 4–5 months and subsequently built and maintained the full production system — including all integrations with OnCore, EMR, and CTMS — within a 14–15 month total engagement, demonstrating both speed and depth in clinical software delivery.
A robust investigational drug service platform requires a reliable server-side framework for business logic, a structured relational database for audit-compliant data storage, a responsive frontend for clinical workflows, and a well-documented API layer for external integrations. Closeloop Technologies built Vestigo on ASP.Net and VB.Net for the backend, SQL for the database, JavaScript and Backbone.js for the frontend, with a comprehensive API suite supporting integration with OnCore, EMR systems, and clinical trial management platforms — a stack chosen for long-term maintainability and clinical-grade reliability.
